CD9 tetraspanin is the only egg membrane protein known to be essential for fertilization. To investigate its role, we have measured, on a unique acrosome reacted sperm brought in contact with an egg, the adhesion probability and strength with a sensitivity of a single molecule attachment. Probing the binding events at different locations of wild-type egg we described different modes of interaction. Here, we show that more gamete adhesion events occur on Cd9 null eggs but that the strongest interaction mode disappears. We propose that sperm–egg fusion is a direct consequence of CD9 controlled sperm–egg adhesion properties. CD9 generates adhesion sites responsible for the strongest of the observed gamete interaction. These strong adhesion sites impose, during the whole interaction lifetime, a tight proximity of the gamete membranes, which is a requirement for fusion to take place. The CD9-induced adhesion sites would be the actual location where fusion occurs. PNAS, 2011
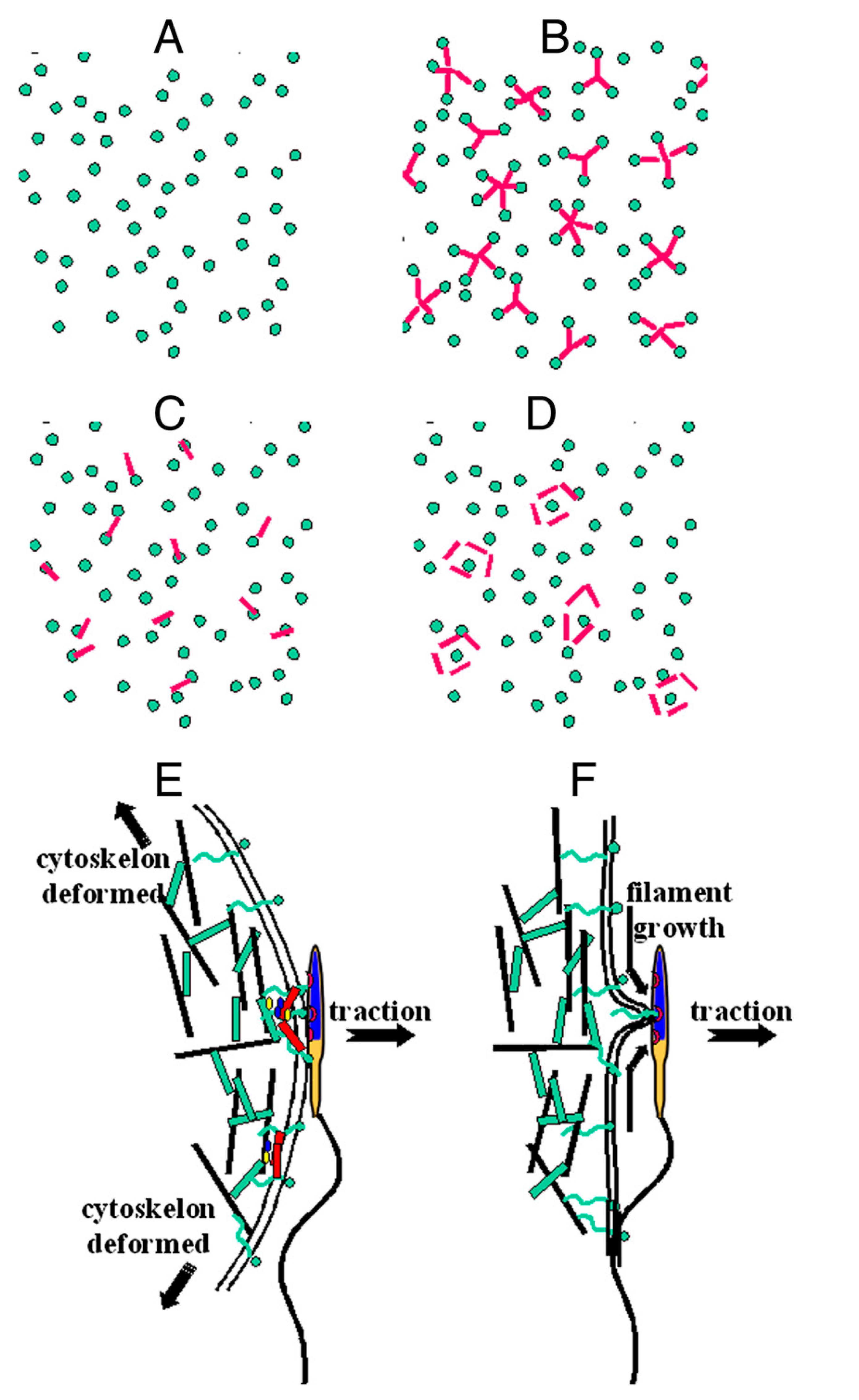
Model of the involvement of CD9 in gamete adhesion and fusion processes. (A) Schematic representation (from top view) of receptor distribution (green dot) on Cd9 null egg microvilli. (B–D) Different possible receptor and CD9 (red rod) distributions that can account for S adhesion at WT membrane (B) CD9 induces clustering of sperm receptors, (C) CD9 associates with a sperm receptor, (D) CD9 are surrounding the sperm receptor. (E) On S adhesion sites, upon traction, sperm head remains stuck to the egg membrane as long as the sperm–egg bond resists. Cytoskeleton gets deformed. Fusion process may occur. (F) Sperm–egg contact takes place on an isolated receptor. Upon traction, as soon as the receptor detaches from the cytoskeleton, a filament starts growing. The surface of contact between the gamete membranes is reduced to 1 point. Membrane fusion is impossible.