Intermediate states during SNAREpin formation prior to membrane fusion and neurotransmission
(click on each corresponding publication to see it)
In each synaptic button, prior to neurotransmission, ~100 synaptic vesicles are closely apposed to the presynaptic plasma membrane in an energy state that makes them ready for fast fusion upon calcium entry. They form a readily releasable pool. It is well established that the zippering of SNARE proteins into a protein complex, called the SNAREpin, provides the fuel for fusion. In addition to this role in fusion, we propose SNAREpins serve as regulators of the distance between synaptic vesicles and the plasma membrane. They participate to control the size and formation of the readily releasable pool.
SNAREpins are made of a four-helix bundle with a cylindrical shape (10-12 nm length and 2-3 nm in diameter) followed by a two-helix coiled coil (~2nm in length). One helix of the four-helix bundle comes from the v-SNARE anchored in the synaptic vesicle membrane and the other three helices are provided by the t-SNARE anchored in the target plasma membrane. They assemble by zippering from their membrane distal to proximal end. It was initially thought that this zippering was continuous. Our past work (Nat. Struct. Mol. Biol. 2007, Nat. Struct. Mol. Biol. 2011) suggested that this model may not be accurate, and that SNAREs may assemble in a step-wise manner. This was subsequently demonstrated with optical tweezers by the Zhang lab (Yale, Science vol 337, pp. 1340-3, 2012). In collaboration with them, we have shown that this is a general feature among the various identified SNAREpins (eLife 2014). Four main transient intermediate states are found (see figure below): 1. Initial assembly: binding of the membrane distal part of the helices (over ~1nm, i.e. a few amino acids); 2. Half zippered state: the four-helix bundle is half zippered; 3. Four helix bundle state: complete zippering of the four-helix bundle; 4. Full zippering: Assembly of the two-helix coiled coil that is connected to the transmembrane domains of both the v- and t-SNAREs.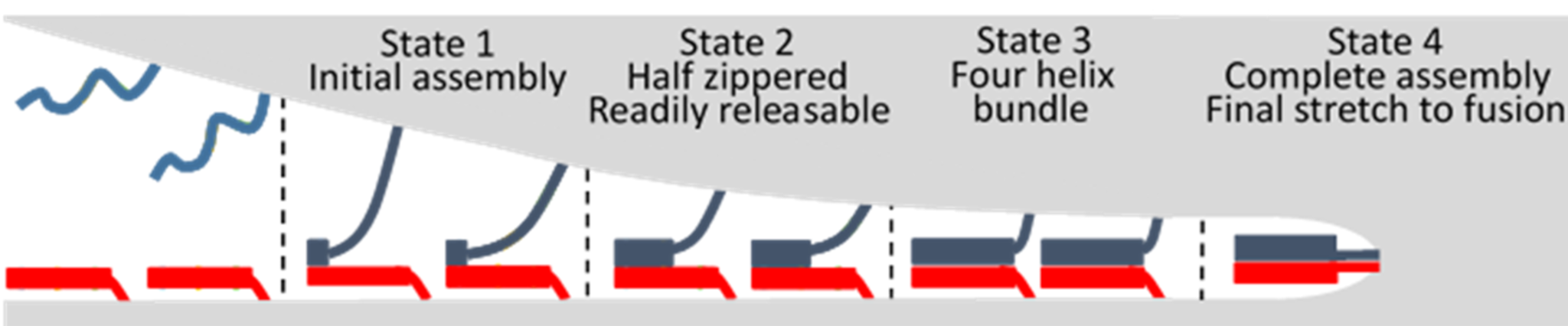
Various intermediate states during SNARE assembly
The transition between disassembled SNAREs and the initial assembly is fast and occurs as soon as the two membranes are close enough for the SNAREs to bump into each other, ~15nm (PNAS 2016 (1)). The physiological role of this initial transition, if any, is unclear. The transition to the half zippered state takes tens of minutes with an activation energy of 35kBT (PNAS 2016 (1), PNAS 2016 (2)). This is the limiting step during the zippering process. We have several threads of evidence that indicate that SNAREpins in the readily releasable pool are in this half-zippered state. Hence this would be the platform from which SNAREpin would start prior to the final zippering towards fusion. The high activation energy may be useful to avoid having too many SNAREpins in the half-zippered state and limit the size of the readily releasable pool. However, it also means that, when needed, this barrier must be actively bypassed to refill the readily releasable pool in less than 100 msec (timescale physiologically observed). This is probably achieved by regulatory factors. The transition to the full four-helix bundle must also be modulated to keep the SNAREpins in the half-zippered state (JACS 2014) before fusion and release it upon calcium signal. Without additional constraint, this transition only requires a few kBT, which is consistent with fast release of the neurotransmitter. Regulatory factors, Synaptotagmin and Complexin, ensure that the SNAREpins do not spontaneously zipper completely (eLife 2015). We believe that the assembly of the two helix coiled coil is the final stretch that actually provides the energy to open and expand the fusion pore (Science 2012, PNAS 2017).
In collaboration with James Rothman (Yale and UCL)